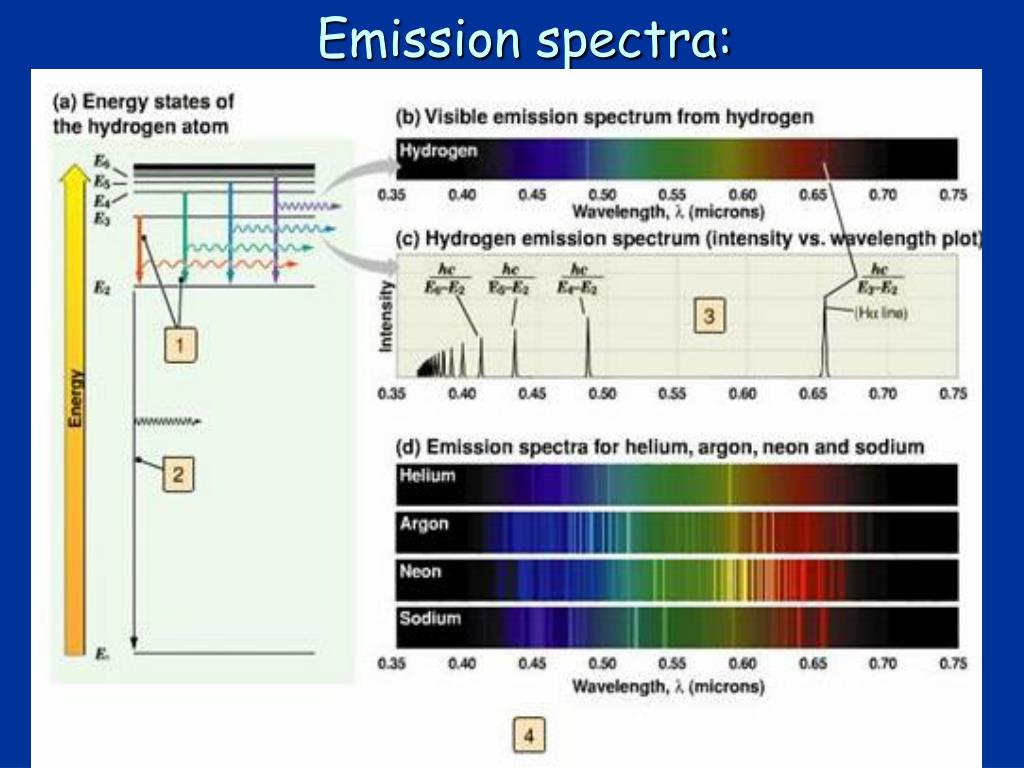
I'm studying emission spectrums at school right now, and my teacher said we're going to do a "flame test" for our astronomy course next week, but I have no idea what that is. temperature steady-state emission spectra were collected using a SPEX. Your instructor must have mentioned to you about Bunson and Kirchhoff who first studied the atomic emission spectrum by using a burner flame and a prism. vivid color and/or luminescence changes in response to specific gases and vapors. As a consequence of plasma inflow the flare emission measure increases. How it works is that when an atom or molecule is heated, it emits radiation which can be seen as a spectrum of bright lines. Such spectra have been measured frequently by the BCS instruments aboard the SMM. Because if an x-ray is an emission spectrum, then why aren't we able to see it?įebruary 4, A flame test is a test where an element is literally held to a flame so that it emits a spectrum of color and by that color, you can determine what that element is. I had heard that an x-ray is also an emission spectrum, but after reading this I'm kind of doubting it. We can see sunlight because it has the right wavelength, but we can't see radio waves. To observe and understand line emission spectra of atoms using gas. To relate these results to the types of electronic transitions occurring in these elements. A line spectrum is like a fingerprint - it can be used to identify the element. To perform flame tests of metal cations in order to observe their characteristic colors, To perform calculations to determine the frequency and energy of the emitted photons. Different elements produce different line spectra. An emission spectrum consists of all the radiations emitted by atoms or molecules, whereas in an absorption spectrum portions of a continuous spectrum (light containing all wavelengths) are missing because they have been absorbed by the medium through which the light has passed the missing wavelengths. an X-ray's wavelength is too short for us to see. Line emission spectra are unique to a particular element. We're not able to see x-rays because not all spectrum is visible. So you can think of x-rays as a single piece that makes up an emission spectrum. 
3: When light from a hydrogen gas discharge tube is passed through a prism, the light is split into four. 
The figure below shows the atomic emission spectrum of hydrogen. When electromagnetic radiation comes together in an arrangement, its called an electromagnetic spectrum. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. Actually an x-ray is not an emission spectrum, its electromagnetic radiation. In an emission spectra electrons are excited to an excited state by thermal or electrical means and then relax back to a lower state and emit a photon of light at a specific energy, which is seen as at a specific wavelength.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
